Cross Contamination Does NOT Make a Good Impression!

A quality infection prevention and control (IPAC) program led by risk management accounts for EVERY little step of EVERY procedure in the dental office analyzed for potential moments of cross contamination. Very simple, if there is even a slight risk, it must be addressed by finding a solution to prevent the cross contamination. Following the hierarchy of controls from the Centre of Disease Control (Figure 1), when a risk presents the first step is to eliminate the hazard or remove the elements associated with the hazard. The second step is to replace the hazard or substitute. When dissecting the steps in impression taking, this article will demonstrate how there is a need to consider tactics of elimination and substitution in our ‘common practice’ verses ‘best practice’.
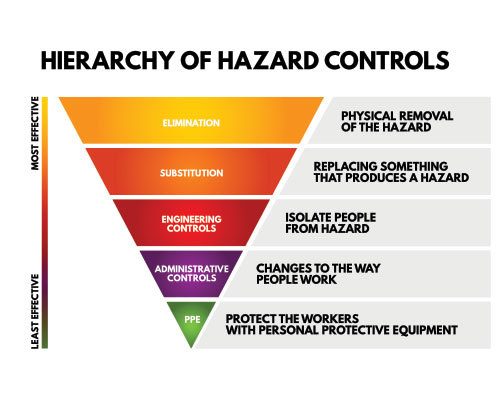
During an IPAC audit, the IPAC Officer of the dental office drafts a list of identified breaches in asepsis and address them one by one determining a solution to mitigate the hazard. One procedure that is often bypassed in this evaluation is impression taking. When the time is taken to break down ALL the steps during impression taking, one may be shocked at the numerous moments of potential IPAC lapses. The break down consists of 7 steps starting from the gathering of armamentarium to the chairside client delivery of the fabrication from the impression.
7 Potential IPAC Lapses With Impression Taking
1. Procedure Set Up
Impression Trays
If the office is using metal reusable trays, are the trays packaged and sterilized properly? In the old ‘common practice’ approaches a dental office had systems that were wonderful for organization but broke every possible IPAC rule. Such an example was a peg board system like one would see in a garage to hang up tools. A series of small hooks where the metal impression trays were all lined up representing various sizes and the arches. The trays were used, flashed sterilized and hooked back up in their reserved position on the board. Another sorting system was bins labelled for the sizes and arches with again flash sterilizing the metal trays. These two examples are better alternatives than the good old ‘cold soak’ system for metal impression trays but do not meet IPAC standards. There needs to be a mention of disposable plastic trays being just that, ‘disposable’, and cannot be sterilized on the plastic cycle or cold soaked. As best practice replaces common practice, dental offices had to adhere to packaging and sterilizing metal trays and abort such setups (note I used the past tense ‘had’ as these IPAC rules came into effect many years ago). Currently, all dental offices, if using metal trays, MUST reprocess them packaged and sterilized under the wrapped/packaged cycle of the sterilizer and open only at point of use
Impression Material
If the material is the alginate canister or the VPS cartridges, is storage such that the canister/cartridges are not cross contaminated? A pandemic or not leaving products/materials on a countertop in an operatory is a no-no. During aerosol generating procedures, which is typically every procedure in a dental office, these containers are subject to contamination by aerosols. If they sit on the countertop, they are fair game to be disinfected after EVERY client. That’s a lot of extra work! So put them away (along with any other clutter)! The top of a cabinet does not count as storage! The reprocessing room does not count as storage either! The reprocessing room operates as stations in a one-way workflow and the corresponding cupboards and drawers must host ONLY what that specific station’s task requires. Are you taking impressions in the reprocessing room? No, so you cannot store the impression material in the reprocessing room!
Mixing Bowls / Spatula & More
The same storage and handling rules apply to the mixing bowls, spatulas, rope wax, wax bite sheets and kidney basins. They too must be in a designated area away from aerosol or direct touch contamination. The reprocessing of mixing bowls and spatulas will be discussed further below.
2. Impression Tray Selection
As one size does not fit all, there is often a trial of a few sizes and widths of trays to find the correct tray for THAT client. So where is the possible cross contamination. Let’s frame the scenario as a ‘case study’ presentation.
Karim is the lead CDA at Smiles for Miles and is very skilled at taking impressions. The set up includes the following:
- 2 alginate bowls
- 1 canister of alginate closed with the alginate pre-dispensed and pre-measured in the alginate bowls
- 1 water measurer
- 1 wooden handle spatula
- Variety of sterilized metal trays
- Kidney basin (metal)
Karim has donned all the required PPE. Mrs. Smith has a narrow palate and Karim is now on the third trial of metal impression trays and is relieved to know it fits! Karim can now finally mix the alginate and load the tray with the spatula. Hurray, Mrs. Smith did great, and the impression is a keeper! On to take the mandibular impression. Karim repeats the exact same process with the exact same armamentarium. Mrs. Smith used the kidney basin only for the first impression with some saliva and pieces of alginate inside the bowl and throws the used tissues inside the bowl. The kidney bowl and alginate trays will be cleaned for the next use. Karim wraps the impressions in a wet paper towel places them in the alginate bowls to transport to the lab for a pour up and follows proper PPE doffing.
Seems typical of a process. Where did Karim go wrong? Here is the domino effect of the scenario having created fomites out of armamentarium that must remain as non-critical in their true allocations within Spaulding’s Classification:
The metal tray was in contact intraoral and is now classified as a semi-critical item as it touched the client’s mucosa. The spatula used to load the tray is scraped along the rim of the metal tray, then used again to scoop up more material in the alginate bowl, contacting the interior of the alginate bowl. The spatula is used again to load another contaminated tray and mix in another alginate bowl, thus contaminating both alginate bowls. The spatula started off as non-critical item and should remain in that designation as per Spaulding’s Classification. Karim did not doff and don between client contact and touching the outside of the alginate bowl, the handle of the spatula and water measurer. The alginate bowl, spatula, water measurer and kidney basin all need to be cleaned and reprocessed. At best the alginate bowl can be disinfected as well as the water measurer, BUT a doff and re-don of gloves would be indicated to lessen the risk of exposure to these high contact items. The water should have been pre-measured and pre-dispensed in a dixie cup to eliminate touching the water measurer with soiled gloved hands. Karim did pre-measure and pre-dispense the alginate but not the water. The wooden spatula should be substituted for a heat tolerable material that can be sterilized. Wood is porous and will harbour microorganisms and cannot be sterilized. The metal kidney basin can be sterilized as it is metal. The alginate bowl was further compromised in being used as a transportation system for the wrapped non disinfected impressions. The impression must be disinfected either in the operatory or in the lab where a controlled spray delivery is ideal to reach all the anatomy of the impression.
Can you see the cross-contamination moments? How can this be mitigated within a proper risk assessment?
The armamentarium requires two spatulas for each mix and possibly four if the clinician is going back to scoop up more material. Another approach is to set up two of each impression trays. Use the first round for trial BUT use a sterilized metal tray or new plastic tray for the actual impression as it will not have come into contact with the client’s mucosa and therefore not be a fomite. The exterior of the mixing bowl and water measurer needs to be handled with a change of gloves or pre-measure and pre-dispense the water in a dixie cup. The impressions need to be transported in a container used for ONLY transportation and then properly disinfected. An alginate bowl’s sole purpose is to mix alginate, not to be a means of transportation. The disinfection of the impression needs to be controlled and follow the directions of the manufacturer for contact time.
3. Mixing Impression Material & Alginate Bowl
See above in the example for the identified moments where the alginate bowl has become a fomite. If the dispensing was via cartridges with VPS material, care needs to be taken for the tip of the VPS cartridge to NOT touch the interior of the tray if the tray has had an intraoral pre-trial fitting.
4. Taking One or More Impression
As per the case study, without the proper armamentarium, going from one impression to multiple continues the cross-contamination domino effect.
5. Operatory Clean Up & Reprocessing
Kidney basins are often overlooked for reprocessing. If it is metal, automatically Spaulding’s Classification states heat tolerable materials must be heat sterilized. If the kidney basin is plastic, it should be lined with a plastic barrier with the barrier removed post use and the kidney basin disinfected. Alginate bowls must be cleaned and disinfected. There should be enough inventory of spatula’s as per the case study as well as having heat tolerable material for the spatulas to withstand sterilization.
6. Travelling / Couriering Impression
Transportation of the impression whether that is physically walking the impression(s) down the hall to the lab in the office or couriering to an external lab for off-site fabrication, there must be a system.
- Internal Lab: There needs to be a transportation system that utilizes a container that can be re-used with proper disinfection.
- External Lab: Would it not be a great project to swab the outside of a lab box to see what type of microorganisms show up? In the moving of the lab box from point A to B, there are many surfaces and hands it touches. The box is a HUGE fomite, so DON’T bring it in the operatory. Leave the box in the lab or another designated area. If there is no other area, take care in writing a protocol and go the step further to collaborate with the lab to ensure elimination or substitution of certain steps to halt the cross contamination. What about the inside of the box? Yet another risk! The mitigation here is to ensure the plastic ‘Ziplock’ bag is not contaminated on the outside.
7. Delivery of Fabrication to Client
All fabricated prostheses must be disinfected prior to client delivery. A meeting with the lab(s) needs to take place to discuss if the lab is sending the fabrication disinfected and what product was used. Many labs in the last few years have been proactive and placed a sticker to share with the dental office the item was indeed disinfected. Open communication with the lab is necessary to inquire about material compatibility with different chemicals used for disinfection. The lab may state a yes or no on certain chemicals depending on the material.
Conclusion
In any IPAC discussion, a proper risk assessment guided by the Hierarchy of Controls and Spaulding’s Classification will never stir any IPAC guru wrong! Even with dental offices having attended countless continuing education on IPAC, there may still be lingering remnants of ‘common practice’ that require a closer look. Striving for that next level of safety is the true goal of any IPAC program and it can be accomplished when a team works together in understanding the hidden fomites and moments of cross contamination. If anything, it's beyond exciting to find these covert elements in our IPAC day to day tasks and work towards solutions that strengthen the offices IPAC program. Cross contamination does not make a good impression, but you can create an ‘impressionable’ IPAC program with your eagle eye approaches of risk management.







