The One-Way Workflow of Reprocessing: Analyzing the Function and Rationale of Each Station.

A dental office reprocessing area must be organized in a one-way workflow to prevent cross-contamination. The one-way workflow is composed of stations that are linked in functionality to one another to finally achieve the end point of reprocessing; sterilization. Technically the one-way workflow endpoint is storage as after sterilization the instruments must be safely stored and distributed to their point of use all while maintaining sterility. Each station can only perform the duties of THAT station and can only host the contents of THAT station’s tasks. In ‘common practice’ vs ‘best practice’ reprocessing rooms, a.k.a. sterile bays, were the hub of the offices' inventory, acting both as a storage and a tray setup area for dispensing and navigating instruments and products. Public health and dental regulatory bodies made it clear many years ago (2017 in Ontario) that a dental office had to mimic the organization of a medical facility where reprocessing and reprocessing ONLY happens in the reprocessing area. This means products like gauze, masks, bibs, saliva ejectors, fluoride, prophy paste, gutta percha, L.A., masks, etc. CAN NOT be stored in the reprocessing area as they have no function in the reprocessing task and their presence in a non-sterile environment poses the risk of being a fomite in cross-contamination.
What are the stations in the reprocessing area and in what order must they appear within the one-way workflow? There are typically seven stations and can vary in the label of the station’s names. The following list is a common approach to naming and sequencing the stations in the one-way workflow.
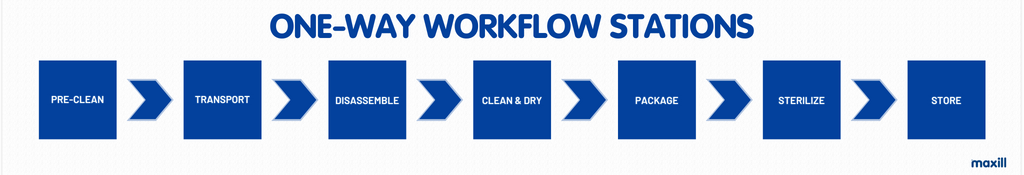
The term ‘one-way workflow’ needs to be defined as the movement of instruments and operators from non-sterile to sterile preventing cross-contamination. Before each station is discussed in detail, it is important to designate which of the stations are part of the non-sterile and sterile designation, as per the image below.
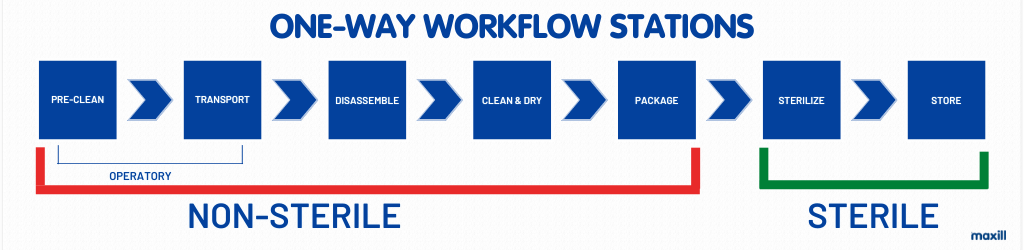
Note how the stations begin in the operatory PRIOR to the entry into the reprocessing room. The pre-clean and transport do not happen in the reprocessing area, they happen in the operatory. If any dental professional performing the task of reprocessing has encountered dry cement on a spatula, dry blood or a chunk of calculus stuck on a scaler, immediately initiating ‘reprocessing’ in the operatory makes so much sense (and a huge favour to the person reprocessing the instruments). Keeping instruments clean at the point of use is part of reprocessing, just as transporting them safely to be reprocessed is.
In the movement from non-sterile to sterile there is a dual moment where at the station of sterilization the sterilizer is involved in being both a non-sterile AND sterile piece of equipment. The dashboard and door of the sterilizer are a fomite. This will be discussed further in the Sterilize Station section. As each station is described, reflect upon the one-way workflow in YOUR environment, does it pose a risk for cross contamination? Does it need to be altered? Taking notes while reading the article will serve as an analysis of your reprocessing area and allow goal setting for positive learning outcomes.
The One-Way Workflow of Reprocessing
Pre-Clean
Pre-Clean at Point of Use
Instruments must be kept in clean working order while being used in client care. This means ensuring gross debris does not dry on instruments. If instruments cannot be reprocessed in a timely manner, they must be immersed or sprayed with an enzymatic cleaner that will not allow the gross debris to dry. If the gross debris dries it may be impossible to remove ALL of it and minute particles may remain on the instrument(s), especially where there is texture, hinges, lumens, cutting edges, etc. The most efficient enzyme pre-cleaning products are those in a gel or foam format sprayed directly on the instruments. The spraying of the instruments with an enzyme cleaner can be performed in the operatory or in the reprocessing area upon ‘drop off’ of a soiled tray of instruments.
Waste Management at Point of Use
This station also includes disposing of waste at the point of use. Do not carry a tray of instruments plus garbage (soiled gauze, used saliva ejector, gloves, etc.) to the reprocessing area. Dispose of waste in the operatories' designated receptacles as identified by the offices' health and safety (H & S) policies of waste disposal. Sharps containers at the point of use are included in the ‘waste’ category in the proper receptacle (sharps container in the operatory). Waste management is a risk management discussion within IPAC and H & S as the person creating the waste and handling the waste should not expose another person to the waste.
Looking for more information on IPAC (Infection Control and Prevention) Resources? Here's a link to our extensive list of PDF checklists and informational articles!
Transport
The transportation of soiled and sharp instruments originates from risk management in H & S. The transportation mechanism must be a closed locking container. Should there ever be a situation where the instruments fall, move, etc. they are secured and the person handling the instruments will not have a sharps injury. The transportation system must be accounted for in either designating ‘soiled’ only transport systems where the containers and lids are stored in a designated ‘non-sterile’ area and only brought out to carry soiled instruments. Another option is to disinfect the transport mechanism and have it serve a dual purpose for carrying sterile instruments (when fully disinfected) into the operatory and non-sterile instruments (post use out of the operatory). The dental office can choose and MUST have a clear written policy and procedural steps so NO ONE CROSS CONTAMINATES. Does a locking instrument cassette ‘count’ as a transportation system? Yes, it can if the instruments' sharp ends do not stick through the cassette’s perforations. Many offices that use cassettes still need some sort of locking tray with a plastic lid or a bin with a secure lid because the ultrasonic insert, slow speed, high speed, etc. are not reprocessed in an ultrasonic bath and/or instrument washer. Typically, in a tray set up the handpieces and motors are not in the cassette and would still need a transportation system (even if they are not sharp, they are soiled and thus pose a risk).
At maxill we carry a wide selection of products to help you safely transport soiled and sharp instruments including:
- Trays with Locking Lids
- Cassettes in Multiple Sizes
- Paper Tray Liners
- Disinfectant Wipes
- Tub, Tub Inserts and Tub Dividers
- And More!
Disassemble
The disassembly task has a few options for the ‘where’ and ‘when’ it is performed. First to be identified is what MUST be unassembled:
- Handpiece motor and contra angle
- Handpiece contra angle, chuck, and head
- Toffelmire retainers and bands
- Oraqix syringe
- L.A. syringe
- Piezo/ultrasonic handpiece sheath
- Bur from handpiece
- Blade from surgical handle
- Needle from syringe
- Etc.
All instruments can be unassembled in the operatory except for the slow speed handpiece (SSH) if it is the style that comes apart with the contra-angle, chuck, and head. Why not disassemble the SSH in the operatory? This is answered by the maintenance task of flushing and lubricating the handpiece prior to sterilization. This task is performed in the reprocessing room with either an automated handpiece docking station, or the good old spray can delivery method. The automated docking station could also be located in the clean and dry station as the handpieces may be disinfected with a lint free wipe prior and post to being mounted in the station. Some offices also like to perform the ‘cleaning’ with a cleaning brush on the handpiece prior to docking. Please read the handpieces MIFU as directions may be to not use a disinfectant wipe on the handpiece, only a lint free towel. So, in essence, your office may choose to encompass the Disassembly and Clean/Dry Stations together, you have choices! Why should all the other instruments listed above be unassembled at point of use? This is going back to the former discussion in this article on sharps containers at point of use and exposing another person to the potential risk of injury. Burs, blades, needles, toffelmire bands as well as ortho wires and ties all need to be disposed of in the sharp’s container in the operatory so no one else handles the sharps.
Clean & Dry
Clean
Any gross debris left on a minuscule area of an instrument will not be sterilized and will be baked on making its removal exceedingly difficult. Cleaning can be accomplished in several ways:
- Manual with a brush
- Mechanical with:
- Ultrasonic bath
- Instrument washer
Manual Brush
For H & S reasons, many years ago in dentistry we were informed to halt the use of a manual brush at the cleaning station for two reasons. The action posed a substantial risk for a sharps injury and the brush needed to be accounted for aseptically. Yes, granted the brush is dirty (extremely filthy actually) thus the reason to not keep adding and adding to the microbe flora. Brushes can be disposed of or sterilized, and the choice will be made by the brush material in which it is manufactured and the MIFU. Many geographical public health regulatory bodies ‘allow’ manual brushing, whereas the dental regulatory bodies discourage its use. Nonetheless, if it is to be performed the brushing must be in a controlled environment brushing only in one direction to reduce spraying and splatter on the operator. Note how the ‘brush’ is handled must be part of the office's IPAC written policies and procedures.
Ultrasonic Bath
The ultrasonic bath functions with cavitation in ‘shaking off’ debris with enzymes specifically selected to break down blood proteins, mucin deposits and overall gross debris. A quality enzymatic solution has the following elements to consider:
- Ecofriendly
- Non-corrosive
- Low pH
- Phosphate free
- Non-caustic
- Low foaming
- Safe to use on: rubber, stainless steel, and plastics
- Simple to measure and dispense
- Multipurpose (an enzymatic cleaner that can be used for cleaning instruments, water lines and suction lines)
There are a variety of ultrasonic baths, and a common issue is offices using a bath too small in size. I see this repeatedly, walking into a ten-plus operatory dental office and the ultrasonic bath is the size of what should fit a 2-op office! This is a high-risk scenario as a small ultrasonic bath will not have the capacity and performance to thoroughly clean. A ‘best practice’ ultrasonic bath needs to have the capability of warming the solution. Think about cleaning your dishes in water that is at room temperature. Not a great outcome for your dishes! The unit should also have a de-gassing button and if it does not, be prepared to de-gas it every time new solution is added. De-gassing removes tiny air particles. Envision the tip of a scaler with microbes and the tip is sitting in one of these tiny air bubbles; it will not get cleaned. The placing, removing, rinsing, and sorting of sharp soiled instruments are four potential moments for an injury and transmission of pathogens to the operator.
Another issue is the dumping of the ‘bacteria soup’ from the ultrasonic bath as in best practice it should be changed at a minimum twice a day. Filled at the start of the day, changed mid-day, and emptied at end of the day. If the bioburden is greater due to more invasive procedures, the solution should be changed post that cleaning load. The tube connected to the back of the ultrasonic bath is a major issue for biofilm and should be considered in an IPAC Program as to when it will be accounted for and when it will be changed. Take the time to have a look at the plastic tube in your office. Most likely it has some pink, grey and black discoloration displaying an intricate matrix of biofilm that is being picked up by the ‘bacteria soup’ every end of the day when it’s dumped in the sink. Biofilm is dangerous to the operator and even more dangerous when its traveling companions are blood proteins and pathogen loaded bioburden.
Instrument Washer
The instrument washer functions with hot water under high pressure to remove debris with the assistance of an enzymatic agent to also break down blood proteins and overall gross debris. If the instrument washer has a disinfecting capacity, even better. The instruments when unloaded from the washer are ‘disinfected,’ not sterile, but much safer to handle than the splashing and spraying from the ‘bacteria soup’ of the ultrasonic bath.
Dry
Drying must be with lint free towels . The laundering of towels must be in a designated area for ‘soiled’ towels only. Staff cannot take the towels home to wash them in their own laundry machine and dry them in their own dryer. This is a cross contamination issue. I am quite sure no one wants to share their laundry machine with these towels … one word and one word only … gross! At this ‘station’ there are decisions to make for efficiency and safety. If the office uses an ultrasonic bath and has not made the investment in an instrument washer, this station will slow down the one-way workflow and elevate the risk of a sharp’s injury. When using an instrument washer, the instruments are clean and dried (disinfected if the washer has that feature)… keyword DRIED. The ultrasonic approach of mechanical cleaning is still acceptable; however, the task has some time management and H & S issues. To accomplish a full removal of the enzymatic cleaning solution the instruments must be rinsed with COPIOUS amounts of water and ALL instruments must be reached (posing a splashing and spraying risk). This means the cassettes should be open to rinse all the tips of the instruments properly. Once rinsed, the drying should be completed in the same approach as silverware at home. Dried one by one as well as the cassette. At home we do not leave the silverware on the plate and roll the silverware dry! An even remotely wet pack/instrument/cassette going into the sterilizer inhibits the sterilization process. A common issue in dental offices is wet packs post sterilization. Well, the pack was never going to have a chance to be fully dry if it arrived in the sterilizer damp!
In the discussion of drying comes the topic of lumens. Any hollow device must be meticulously cleaned and fully dried. The MIFU of the lumen must be consulted to determine the steps for either cleaning with an ultrasonic bath or cleaning with an instrument washer. Many MIFU of lumens recommends the instrument washer if the lumen is wide enough for the water pressure to enter. Otherwise, the basic steps for lumen reprocessing are as follows:
- Clean with soap and water
- Rinse with sterile water with a monojet syringe
- Dry with dry compressed air
- Package
Can you see why many offices are using disposable air water syringe tips? How many reprocessing areas at the drying station have a dry air compressor? I personally know of only one, where I teach at Fanshawe College as it was purposely added in the design of the decontamination area of the reprocessing room. Next to a DUWL (dental unit water line), one of the smallest lumens in a dental office is the narrow tubing of the air water syringe and not only is it narrow but it has an angle. Can you envision the image under an electron microscope at the angle junction? Disposable hollow devices, such as air water syringe tips, are a best practice approach to meet IPAC standards due to the fomite risk of reprocessing lumens.
Packaging
As one station works with the previous and next station, the packaging is fully dependent on having dry instruments/cassettes. As stated above a wet pack going in will be a wet pack coming out. So, the first ‘rule’ of packaging is to ensure complete dryness. Years ago, instruments were ‘flashed’ sterilized, a.k.a. sterilized unwrapped, unpackaged and the one-way workflow did not have a ‘package’ station. The moment an instrument meets the air when the door of the sterilizer opens, it is considered non-sterile. For this reason, sterility must be ensured with closed packaging that will not allow microbes to enter. A wet pack is a semi-permeable membrane and as soon as that sterilizer door pops open, microbes have an opportunity to cross a wet barrier.
Instruments are either packed in a sterilization pouch or wrapped in sterilization paper if a cassette. The key highlight of the packaging is to ensure the pack/wrap is fully sealed. This means the pouches are to be sealed precisely on the folded line of the adhesive and nowhere else. This means the cassette wrap is to be fully secured with autoclave tape (external Type I chemical indicator). Pouches must have an internal Type IV chemical indicator and external Type I. For the discussion on placing a Type V in every pack please read the following blog article that offers in specific detail the rationale for the two roles of the Type V chemical indicator : load leader in a process challenging device (PCD) and package leader in packs. Also read the article on the purpose of a PCD.
What other task is performed at the packaging station? Labeling! Again, as in packaging, years ago, labeling was not performed. Labeling is a part of an IPAC Program’s Instrument Recall Process, in the event of a sterilization failure. Packages are labeled with the following:
- Date Sterilized (not packaged)
- Sterilizer
- Load #
- Contents (if not seen visually)
- Initials (of person loading the sterilizer)
Why is it the date of sterilization and the person sterilizing, versus the date packaged and the person doing the packaging? This can be answered with two questions … Are we performing quality assurance testing on the sterilizer or the packaging? If there is a recall of instruments, is it dependent on how and who packed the instruments or on how and who sterilized the instruments? Correct its all dependent on the sterilizing process!
Both packaging and labeling are the most consuming tasks of the one-way workflow. How can an office make the task more efficient and accurate? Label guns, digital barcode labels, instrument tracking/tracing and quality assurance activity logging for public safety and proof of office compliance. Label guns operate similarly to the pricing labeler in a grocery store. It is an efficient and neatly printed approach to labeling the information required on every package. For the offices that have paper charts, the dual adhesive labels can be placed in the client charts serving the requirements for a tracking system within the office's IPAC Program’s Instrument Recall Process policy. This will suffice the requirements for both labeling and tracing, however, will not be as efficient in an IPAC lapse for quick retrieval of what instruments were used on what client, like what a digital barcode logger can deliver. For this reason, maxill has been working for the last year on developing and creating a revolutionary digital logger that has been programmed to parallel IPAC quality assurance sterilization monitoring standards. The system is written by dental professionals for dental professionals to take it one step further than only verification and logging by ensuring guidance and direction with prompting screens in the event of a sterilization failure. Working with digital scanners and unique barcodes ensures a competent system that eliminates the risk of error preventing the use of a non-sterile package on a client. Having digital centralized data for tracing and tracking also means in the event of an IPAC lapse the sterilizer and load number are searched and a list of clients attached to that pack(s) is populated. NO GUESSING! Needless to say, this is an astronomical safeguard approach for the dental office. For more information, please follow this link: https://www.maxill.com/ca/steri-sox-st .
In retrospect, how odd is it in past years how the packaging station never existed? This means dental offices had ZERO approaches to tracking and tracing instrument use within an IPAC lapse and proving to the public they did not infect a client. On the public side of the fence, a client could not have been notified precisely of an IPAC lapse. Such notification would have been a broad time span and may have included clients not involved in the lapse, what a nightmare! What a risk to take!!!! No wonder the ‘Packaging & Labelling Station’ was introduced along with all its important tasks.
Sterilize
Sterilization does depend on all the other stations in the one-way workflow. As discussed above, there are many moments throughout the one-way workflow that will interfere in proper sterilization. Sterilization also depends on the operator to have been properly trained on the specific model and type of sterilizers in the office. This knowledge involves:
- Proper loading of the sterilizer
- Regular maintenance of the sterilizer
- Troubleshooting potential issues
- Analysing error codes from the dashboard
- Understanding quality assurance testing for sterilization monitoring
EVERY member of the dental team needs to complete IPAC training in reprocessing. Why every member? Why not just the person doing the reprocessing? Granted 100% for the person(s) doing the reprocessing but think of the client asking IPAC reprocessing questions to the administrative team after their appointment or before their appointment over the phone. Would you like your administrative team to be able to answer these questions promptly and accurately? A hesitation in these teachable moments could create uncertainty for the client and impact the office negatively.
It was mentioned earlier, that in the movement from non-sterile to sterile the sterilizer is involved in being both a non-sterile AND sterile piece of equipment. The dashboard and door of the sterilizer is a fomite. This is why an office IPAC manual needs clear instruction on what PPE to wear when loading the sterilizer, closing the door, selecting the cycle and pushing start. Aside from a gown, mask, eyewear and face shield, Ideally, the approach would be to load the sterilizer with utility gloves, doff the gloves, hand hygiene, and operate the sterilizer's dashboard. A step of donning gloves could be added after hand hygiene, should the office want this as their protocol. Now the sterilizer has completed the cycle and switches its hat to sterile. In this task the office needs to decide will the controls on the dashboard, the door and the instruments be handled with clean hands (having just performed proper hand hygiene) or with hand hygiene followed by donning gloves. THIS MUST BE IDENTIFIED IN YOUR IPAC MANUAL! If not identified, can you see the risk for cross contamination?
Store
Unlike Vegas, what happens in the reprocessing area, does not stay in the reprocessing area! The only allowable storage area for sterile instruments in the reprocessing one-way workflow is POST sterilization. Not on top or below the sterilizer. The top of the sterilizer would run the risk of having a moisture issue, especially in the sterilizers where steam is vented from the top of the sterilizer. The bottom cupboards or drawers of the sterilizer run the risk of contamination when dirty instruments are loaded into the sterilizer. If a cupboard is not present AFTER the sterilizer to store instruments, they must be distributed to the operatories. Within the operatories, there must be a designated cupboard or drawer for sterile instruments and cannot be housed with sundries, products, etc. So, in some offices, the last station of storage can actually be the operatory.
Other Considerations in the One-Way Workflow
Is there anything else applicable in the one-way workflow? Yes, three more!
- PPE
- Physical Barriers
- Eye Wash Stations
PPE
Every station needs the proper PPE! The selection of PPE includes:
- Gowns
- Utility puncture resistant gloves
- Regular client care gloves
- Safety glasses
- Face shields
- Masks (remember pick the mask for the task!)
The decision of what to wear at what station is based on a proper risk triage considering the following:
- Risk of splatter/spatter/spraying
- Risk of contamination on uniform from reprocessing area to other areas of office
- Risk from aerosols
- Risk of a sharp’s injury
- Risk of a hazard injury (burn, chemical, fire, etc.).
A ‘common practice’ versus ‘best practice’ is to reprocess instruments wearing client care gloves. Hooray on wearing gloves, but they are the wrong gloves. Reprocessing MUST be performed with puncture resistant gloves.
Physical Barriers
Since at one-point non-sterile meets sterile, there needs to be a physical barrier to separate the two. As an update from the previous image, see the image below for the complete one-way workflow with physical barriers.
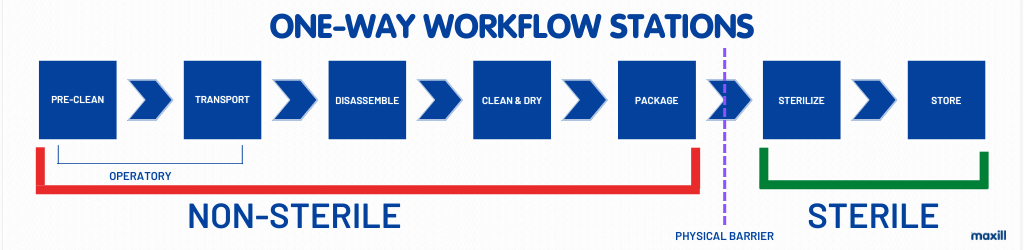
Eye Wash Station
What if the reprocessing has a plumbed in eye station, where should it be in the one-way workflow? It could be located at the end past the sterilizer or past the storage and would need its own physical partitions. Where should the eyewash station NOT be located? On the faucet in the ‘dirty sink’ where the instruments are rinsed! Adding a plumbed eye wash station is not a new regulation for dental offices. It is one that creates confusion as many offices do not have the space and have to be highly creative for their location to meet H & S guidelines as well as IPAC. maxill offers several eyewash station configurations.
Conclusion
How did you do in assessing YOUR one-way workflow? Do you have any enhancements to make? Were you already performing at a high functioning one-way workflow? In many IPAC discussions, my favourite line is ‘we don’t know what we don’t know.’ Caution, however, once we know then we must act on it and can set goals towards changes. These moments of reflection are prime examples of explementary professional growth.
It is clear to see how each station is dependent on each other. Operating in anything but a one-way workflow poses an IPAC risk. Defining the details of each station is important to connect the station and understand how each serves a specific role and why that role is needed to keep moving in the one-way direction. Stay tuned for part two of the one-way workflow where quality assurance tasks will be broken down per station to ensure proper equipment testing, maintenance, and logging. maxill is here to provide products for your one-way workflow reprocessing. Most important, maxill is here to support your critical thinking of the how and why of your IPAC evidenced based decision-making processes.
Resources
- PH Region of Waterloo. Dental IPAC Poster. https://www.regionofwaterloo.ca/en/health-andwellness/resources/Documents/Dental_IPAC_Poster.pdf.