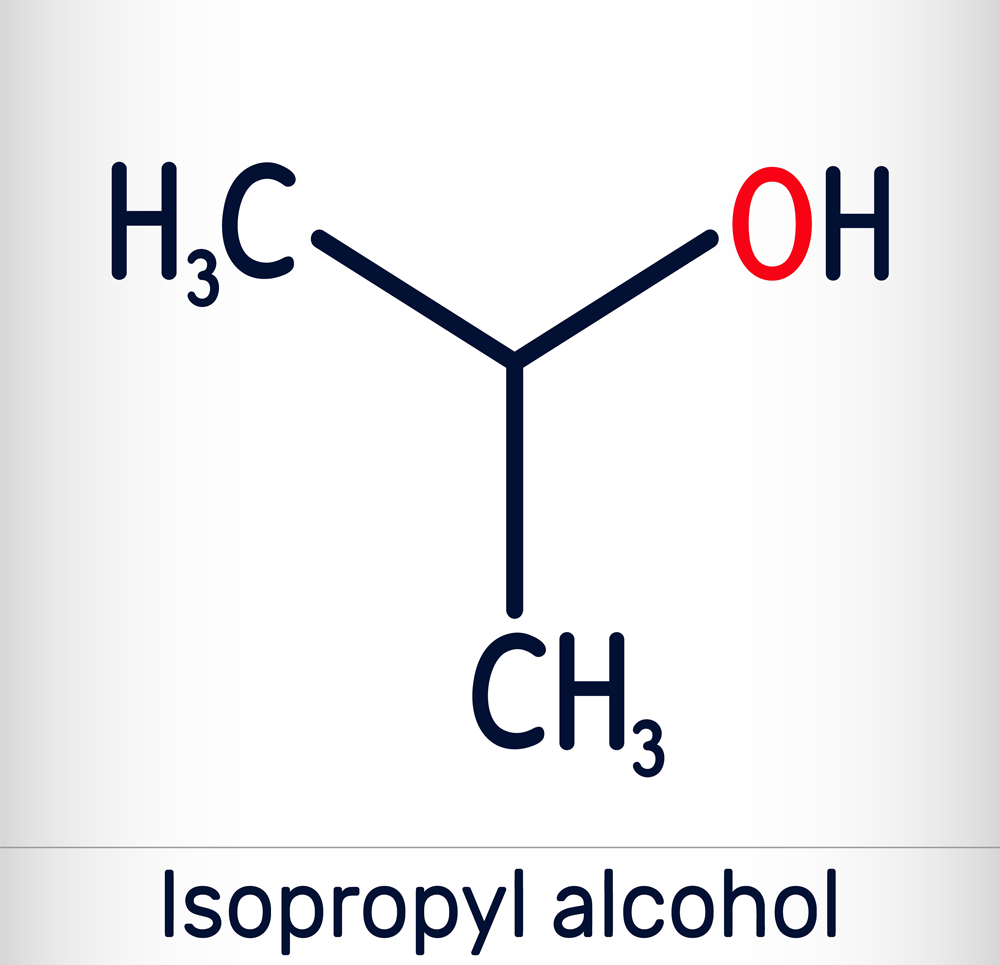
What is Isopropyl Alcohol?
Infection control is an indispensable part of hospital environments and medical settings.
Regular disinfection ensures that working surfaces are kept clean, and potential pathogens like bacteria, viruses, and fungi are eliminated, thus reducing the risk of passing on contamination between patients and between patients and the clinic or hospital staff.
Several types of disinfectants are available in the market, each with a different active component in variable ratios and concentrations.
The choice of disinfectant depends on the spectrum of antimicrobial activity, access, safety factors, handling factors, contact time, and ease of use.
The names Isopropyl Alcohol (IPA) and Isopropanol are synonymous.
IPA is one of the two alcohols used as a disinfecting agent in dental office, hospitals, and medical laboratory facility sanitation.
It is popular in cleanrooms because of its unique chemical composition that works effectively to destroy most bacteria, fungi and viruses.
However, IPA cannot kill bacterial spores, so it is not considered a sterilizing agent. 99% concentration is known as pure isopropanol.
When mixed with a percentage of purified water by volume, we get different grades and concentrations of isopropyl alcohol.
For example, when combined with 10 percent or 30 percent of purified water, we get 90% IPA and 70% IPA, respectively.
Isopropyl alcohol concentrations between 60-90% are frequently used in laboratories owing to their bactericidal, viricidal, and fungicidal properties (CDC).
However, with percentages below 50% and above 91% isopropyl alcohol concentrations, the potency drops rapidly, and they are ineffective as disinfectants.
Another commonly used disinfectant is ethanol or ethyl alcohol, to learn more about the differences between ethanol and isopropyl, see our post Ethanol Alcohol vs Isopropyl Alcohol.
This post will discuss different isopropanol grades, their features and uses.

Are Isopropyl Alcohol and Rubbing Alcohol the same?
The terms isopropyl alcohol and rubbing alcohol are used interchangeably in everyday speech, but in reality, they are two different products.
Rubbing alcohol is an antiseptic usually made from isopropyl alcohol.
According to the CDC, rubbing alcohol is composed of 70% isopropanol and 30% water.
More technically, it may contain between 68% to 72% of isopropanol.
Rubbing alcohol has additives like perfumes, essential oils and stabilizers mixed in with water, which distinguishes it from pure isopropanol.
In addition, it also has added denaturants to make it unfit for human consumption.
Rubbing alcohol is also available in a 97.5- 100% denatured ethyl alcohol/water mixture.
This industrial variety of >91% rubbing alcohol is cheaper and more readily available.
In addition, since ethyl alcohol or ethanol is the principal constituent in alcoholic drinks, it is more palatable for human consumption and less toxic than isopropyl rubbing alcohol.
Are isopropyl alcohol wipes effective for disinfection in dental office sanitation?
Industry-standard isopropyl alcohol wipes, also known as disinfectant wipes are very effective at killing microbes in dental environments.
However, bacteria found on dental practice surfaces are more resistant to antibacterial agents than microbes in free states (like water).
Therefore, surface cleaning with a detergent is required before using a disinfectant.
Subsequently, both the double wiping method and the physical action of wiping are vital for proper disinfection.
Isopropyl and ethanol alcohol wipes are an excellent option in medical and dental spaces because of their convenience of use, disposable nature and need less need for storage space than liquid containers or sprays.
In addition, they contain precise quantities of alcohol required for disinfection and prevent waste from occurring.
They offer an efficient mix of design and application.
Pre-saturated alcohol wipes are available in plastic bags, containers or pouches where the wipes can be pulled individually from the top.
They are also available in individual packets, which are ideal for use because of easy storage and access.
High concentrations (>91%) of isopropyl alcohol are used to wipe down electronic equipment parts because of their quick evaporation and high purity.
Is 70% IPA more potent at killing bacteria than 90% IPA?
Lower concentrations of IPA do not exhibit biocidal properties.
Higher concentrations do not show any improved disinfection either.
IPA 70% hits the sweet spot when it comes to disinfection, especially in dental settings.
Several components in the chemical composition contribute to this efficacy.
The number one reason IPA 70% is so effective in killing bacteria is because it is diluted with water.
Water plays a crucial role in the bacterial protein denaturation process, promoting cell wall damage, increased membrane permeability, protein coagulation and eventual cell death.
In addition, the presence of water decreases the evaporation time, thus increasing contact between the IPA and the bacterial cells and causing lysis.
In contrast, 90% alcohol evaporates before it can penetrate the bacterial cell walls. Therefore, IPA 90% is less effective as a disinfectant when compared to IPA 70%.
A concentration of IPA greater than 91% causes bacterial cell wall proteins to coagulate immediately, giving insufficient time for the alcohol to move into the cell and kill the pathogen.
The coagulated outer membrane forms a protective layer around the microbial cell.
Ironically, it makes the bacteria even more impenetrable. Thus, the surface injury shelters the organism from the IPA, rendering it weak and ineffective for use.
Common uses of Isopropyl Alcohol
Isopropanol is quick-acting and has a broad range of antimicrobial properties.
For this reason, it is often used to disinfect oral thermometers, medical instruments, and external surfaces for sanitizing dental office equipment in clinics and laboratories.
IPA can be substituted with ethanol for use in wet labs and dental facilities.
Furthermore, isopropyl and ethanol can be combined to increase the efficacy of the disinfecting product since IPA is more effective against bacteria and ethanol is more effective against viruses.
Conclusion
maxill® is a great source for disinfectants, including alcohol IPA based.
Just take a look at our website and you'll notice that prices are amazing!
That's because we manufacture pretty much everything we sell, so buying from us means you'll avoid middleman costs.