Sterilization Pouches – Summary of Best Practices

Dental professionals spend numerous hours a day reprocessing dental instruments.
We open the box of sterilization pouches, fill them with instruments, and send them to the heat and pressure of our autoclaves.
How much thought do we put into that little plastic and paper bag?
The United States Food and Drug Administration (FDA) classifies sterilization pouches as a Class II medical device.
The agency considers a sterilization wrap (pack, sterilization wrapper, pouch, or another accessory) a device intended to enclose another medical device to be sterilized by a health care provider.
It is intended to allow sterilization of the enclosed medical device and maintain the enclosed device's sterility until used.
It is also important to note that if the integrity of the packaging has been compromised such as being ripped or wet, the items must be reprocessed as they are no longer considered sterile.
The use of sterilization pouches that are no longer considered sterile is considered an ‘off label’ use therefore not recommended by the FDA.1
RELATED: Sterilization Pouches and Expiry Dates
How Do Sterilization Pouches Work?
Dental sterilization pouches come in assorted sizes to accommodate our dental instruments and devices.
The best practice is following the selected product manufacturer's instructions for use (IFU).
Pouches are manufactured for specific sterilization methods, including pressurized steam, dry heat, or chemical vapor.
Most dental offices use pressurized steam methods.
Following the proper cleaning, packaging, and sterilization procedures will protect your patients from cross-contamination.
A self-sealing sterilization pouch consists of two main parts: medical-grade paper and a transparent plastic film, held together by a heat seal or an adhesive.
The structure of the paper will allow penetration of the chemical, vapor, or steam.
It promotes easy drying, and after complete drying, the pores in the paper close to block any microbe from re-entering the sterilized pouch.
The plastic film enables examination of the contents and a clear view of the color-changing process indicators.
 Loading Sterilization Pouches
Loading Sterilization Pouches
It is a recommendation from the CDC (Centers for Disease Control) that we follow the packaging guidelines provided by the Association for the Advancement of Medical Instrumentation (AAMI). Additional considerations from autoclave and instrument manufacturers recommend separating instruments during the sterilization cycle. Separate metal instruments (e.g., stainless steel, carbon steel, etc.) to avoid oxidation of unlike metals during sterilization. It is also important to wear the appropriate PPE.
Best Practice for Loading Sterilization Pouches
- Hinged instruments should be opened and unlocked.
- Disassemble items with removable parts.
- Position items with concave surfaces to make for easy draining.
- Position heavy instruments and devices as not to damage delicate items.
- Instruments must be prepared for packaging in a manner that will allow adequate airflow and steam penetration (e.g., avoid overfilling packages).
- An internal chemical indicator should be placed in every package, and an external chemical indicator (e.g., chemical indicator tape) should be used when the internal indicator cannot be seen from outside the package.
- Self-sealing sterilization pouches should be sealed using the built-in adhesive strip folded on the perforated lines to ensure an accurate airtight seal.
- As always, follow the device/instrument manufacturer's instructions for use.
- Each package must be labeled on the transparent side of the pouch (taking care not to block the breathable area of the package) with:
- The sterilizer used
- Cycle or load number
- Date of sterilization
- Expiration date (if using shelf-life practices)
- Initials of the person who packaged the instruments
Preventing the Overloading of Sterilization Pouches
The general rule for loading instruments into sterilization bags is to make sure the loading allows for adequate airflow and steam penetration.
Choosing the right size of sterilization pouch helps to prevent overloading.
If a pouch is too large, there will be excessive movement of the instruments.
This may result in taring or punctures of the autoclave bags.
If the pouch is too small, that may prevent adequate steam penetration and air removal.
This could also lead to taring our punctures in the sterilization pouch and may not allow for proper drying.
Best practice states that there should be a minimum of 1” of space between the top of the instrument and the seal of the pouch and that instruments should never be layered.
Even with all these guidelines being followed, human interpretation of the guidelines is still a factor.
What is considered overloading a pouch to one dental professional, might not be to another.
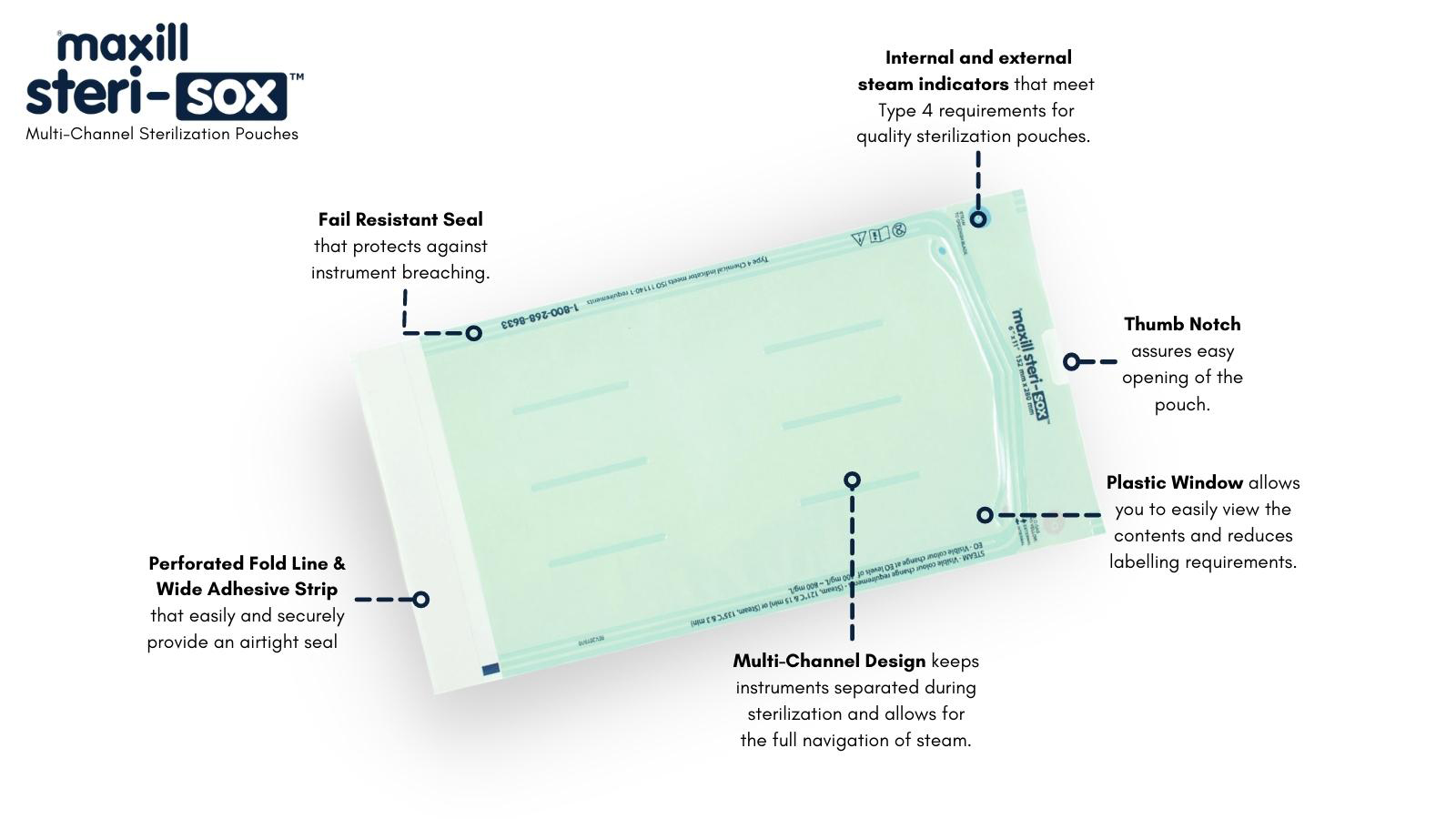
Luckily, new varieties of sterilization pouches, like maxill's new Steri-Sox Multi-Channel Sterilization Pouches, incorporate individual channels within the pouch.
These channels ensure separation and assure that all surfaces of the item will contact the sterilizing component, whether it is steam or vapor.
Internal (Type 4) and external (Type 1) steam indicators meet Type 4 requirements.
Our multi-channel sterilization pouches are available in 2,4, and 6 channels.
Storing & Maintaining Sterility of Sterilized Instruments
Although some dental offices continue to use shelf-life practices (first in, first out), most dental offices have switched to event-related practices.
Under event-related practices, if the integrity of the package has been kept during storage, peel sterilization pouches will keep the sterility of the enclosed device until opened.
Maintaining sterility is achieved if the pouches stay untouched until completely dried.
If handled before complete drying, the paper side of the pouch can easily tear or rip.
Additionally, microorganisms and pathogens can be absorbed and penetrate the inside of the pouch through the wet paper side, breaching the sterile environment by appropriate drying time.2
Best Practice for Storing Sterilized Instruments:
- Store sterile instruments in locations where they will not become wet.
- Avoid excessive handling of pouches as the probability of contamination increases with increased handling2.
- While pouches will maintain the sterility of the enclosed device until opened, the expiry dates that some manufacturers put on their packaging products must also be followed.
- Extremes of temperature and humidity must be avoided.
- Instruments/devices must always be stored at a temperature of 18°C to 23°C and a humidity level of 30% to 60%.
- Prior to use, sterile packages/bags must be checked to ensure that the sterility has not been compromised.
To Sum it Up:
Dental professionals spend a lot of time reprocessing dental instruments.
Careful attention to detail when reprocessing dental instruments and devices will ensure the safety and health of your patients.
Even so, individual interpretation of guidelines and human error can jeopardize the integrity of sterilization.
Manufacturers are working to develop ways to neutralize these components.
Such as supplying more detailed guidelines in the products IFU and creating products to make the sterilization process easier and streamlined.
For example, maxill’s multi-channel sterilization pouches prevent overloading.
Sources:
- US Food & Drug Administration Code of Federal Regulations Title 21, Volume 8, 21CFR880.6850. Revised April 1, 2019.
- "Sterilizing Practices." Centers for Disease Control and Prevention, September 18, 2016, https://www.cdc.gov/infectioncontrol/guidelines/disinfection/sterilization/sterilizing-practices.html.
- “Infection Prevention and Control (IPAC) Guidelines.” Cdho.org. 2022. Available at: https://www.cdho.org/docs/default-source/pdfs/reference/guidelines/cdho-ipac-guidelines.pdf